In brief: Research suggests that full spectrum CBD — which retains terpenes, minor cannabinoids, and trace THC alongside CBD — may offer advantages over CBD isolate. A 2023 pharmacokinetic study found that full spectrum products deliver 12–21% more CBD to the bloodstream than isolate (Berthold et al., 2023), and a 2015 study showed that a whole plant extract maintained a predictable dose-response where purified CBD lost efficacy at higher doses (Gallily et al., 2015). The reasons for this difference involve some of the most interesting findings in recent cannabinoid research.
This article is based on published scientific research and does not constitute medical advice. All findings are attributed to their original sources.
Purified CBD should, in theory, be the most reliable form of the compound — precise, standardised, predictable. A 2015 study published in Pharmacology & Pharmacy found something unexpected: purified CBD followed a bell-shaped dose-response curve, meaning it was effective only within a narrow dosage window and lost its anti-inflammatory effect at both lower and higher doses. A CBD-rich whole plant extract, tested under the same conditions, behaved entirely differently — its effectiveness increased steadily with dose, following the linear pattern researchers need for reliable clinical use (Gallily et al., 2015).
That finding — that the extract overcame a fundamental pharmacological limitation of the isolated compound — is one of the reasons the full spectrum CBD concept matters. It is not simply a marketing label. Over the past decade, evidence from pharmacokinetic research, receptor-level studies, and clinical observation has converged on the same conclusion: the full chemical profile of the cannabis plant may offer advantages that purified CBD alone does not. This article reflects research available as of March 2026.
What this article covers:
- Full spectrum CBD showed 12–21% higher oral bioavailability than CBD isolate in a 2023 pharmacokinetic study — trace THC increased CBD’s intestinal absorption (Berthold et al., 2023)
- Purified CBD follows a bell-shaped dose-response (effective only at a narrow dose range); a CBD-rich whole plant extract showed a linear, predictable dose-response in the same study conditions (Gallily et al., 2015)
- CBD-rich extracts required approximately 4× lower doses than purified CBD to achieve comparable improvement rates in epilepsy patients (Pamplona et al., 2018)
- Why a 2015 study on dose-response curves may be the most practically important finding for anyone choosing a CBD product — and what recent critical reviews say about the limits of this evidence
- After reading, you will understand the scientific basis for choosing between full spectrum, broad spectrum, and isolate CBD
What Actually Is Full Spectrum CBD — and How Is It Different from Isolate?
The dose-response finding from 2015 raised a question that has only become more pressing as the CBD market has grown: if the whole extract behaves differently from the isolated compound, what exactly is in the extract that makes the difference?
To answer that, it helps to understand what “full spectrum” actually means — and how it compares to the two other forms of CBD available on the market.
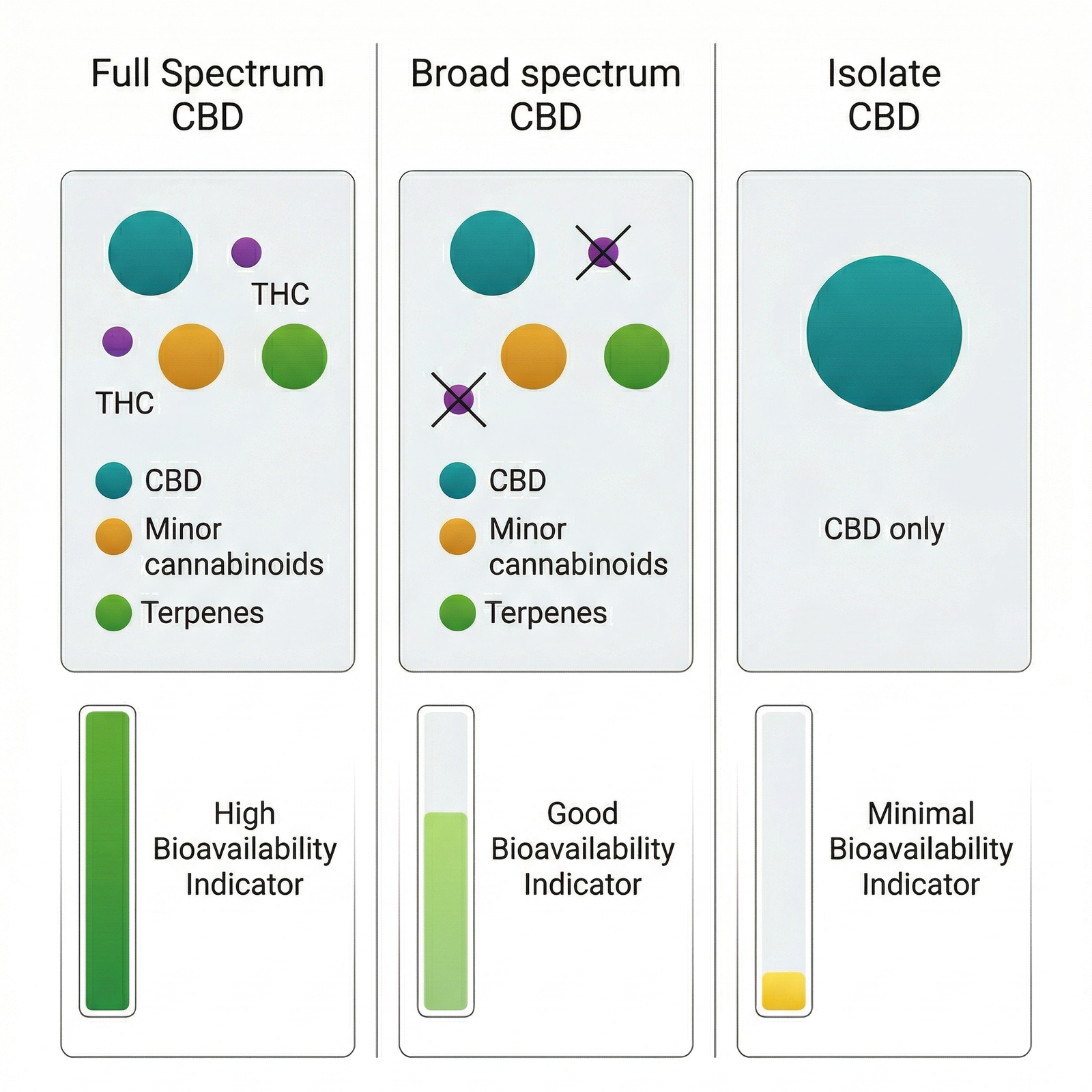
CBD isolate is cannabidiol in its purest form — typically ≥98% purity, with all other plant compounds removed. It is a crystalline powder that contains one active compound: CBD. Epidyolex, the only CBD medicine authorised by the European Medicines Agency, is based on highly purified CBD. This gives isolate a clear advantage in clinical research: precise dosing and reproducibility.
Full spectrum CBD retains the broader chemical profile of the cannabis plant. A typical full spectrum extract contains CBD as the primary cannabinoid, along with trace amounts of THC (below 0.2% in the EU), minor cannabinoids such as CBG, CBC, CBN, and CBDV, and a range of terpenoids — β-caryophyllene, limonene, linalool, myrcene, and α-pinene among the most common. It also contains flavonoids and other plant compounds. The proportions vary by cultivar and extraction method.
Broad spectrum CBD sits between the two: it undergoes additional processing to remove THC while attempting to retain the other cannabinoids and terpenes. The result is a compromise — some of the chemical diversity of full spectrum, without the trace THC that concerns some consumers or that complicates regulatory status.
Why does this compositional difference matter? Because each of these compounds has its own pharmacological profile. β-Caryophyllene, the most abundant terpenoid in decarboxylated cannabis extracts, is a confirmed selective agonist at the CB2 cannabinoid receptor (Gertsch et al., 2008). A 2023 study published in Biochemical Pharmacology demonstrated that cannabis terpenes activate CB1 receptors at 10–50% of THC’s potency, and several synergize with THC beyond additive effects (Raz et al., 2023). In a full spectrum extract, these compounds are present alongside CBD. In an isolate, they are absent entirely.
That distinction is not trivial. It is the difference between one active compound acting alone and multiple compounds acting on multiple biological targets simultaneously. For a deeper look at the receptor-level evidence, see our full guide to the entourage effect.
Key takeaway: Full spectrum CBD is not simply CBD with impurities — it is a multi-compound extract containing cannabinoids and terpenoids that each have their own demonstrated pharmacological activity, and whose combined effects may differ meaningfully from CBD alone.
Does Full Spectrum CBD Actually Work Better? What the Evidence Shows
The compositional argument is logical. But science requires more than logic — it requires data. Three key studies, each addressing a different dimension of the question, form the current evidence base for the full spectrum advantage.
The Bell-Shaped Dose Response (Gallily et al., 2015)
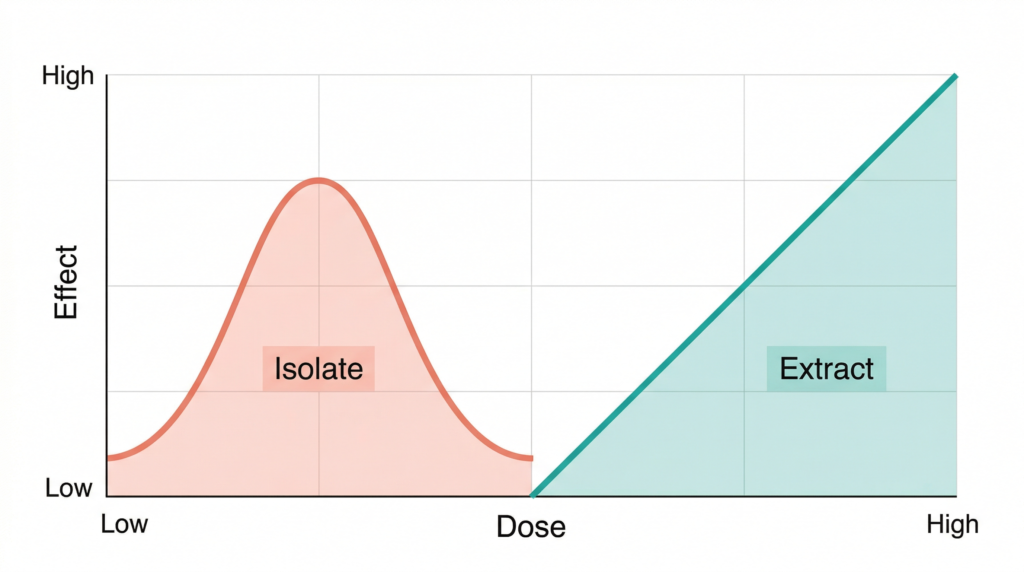
The 2015 study from the Lautenberg Centre at the Hebrew University of Jerusalem is arguably the most practically important finding for anyone evaluating CBD products. The researchers compared the anti-inflammatory and pain-relieving effects of purified CBD against a standardised extract from Cannabis clone 202 — a cultivar highly enriched in CBD with minimal THC.
Purified CBD produced a bell-shaped dose-response curve. At 5 mg/kg, it achieved a 43% reduction in TNF-α production. But at both lower doses (1 mg/kg) and higher doses (25 and 50 mg/kg), the anti-inflammatory effect disappeared. The extract behaved entirely differently: its effectiveness increased consistently with dose, reaching a 57% reduction at 50 mg/kg and continuing to improve at 150 mg/kg (Gallily et al., 2015).
What does a bell-shaped curve mean in practice? It means that with purified CBD, finding the right dose is essential — too little does nothing, and too much also does nothing. The whole plant extract removed this constraint, making dosing far more forgiving and predictable. The researchers concluded that other components in the extract likely synergize with CBD to overcome this limitation.
Bioavailability — How Much Actually Gets Absorbed? (Berthold et al., 2023)
Even if a compound is effective, it matters how much of it reaches your bloodstream after you swallow it. A 2023 pharmacokinetic study published in the European Journal of Drug Metabolism and Pharmacokinetics directly compared the oral bioavailability of commercially available CBD isolate, broad-spectrum, and full-spectrum products.
The full-spectrum product — containing 3.1% CBD along with 0.2% THC, terpenes, and minor cannabinoids — produced 12% higher oral bioavailability in one group and 21% higher in another, compared to both isolate and broad-spectrum products. In vitro permeability assays confirmed the mechanism: the presence of trace THC increased CBD’s intestinal permeability while reducing its efflux through the gut wall (Berthold et al., 2023).
This is a pharmacokinetic finding, not a therapeutic one — it tells us about absorption, not about what happens after absorption. But it has direct practical implications. If full spectrum CBD delivers more compound to the bloodstream per milligram consumed, a lower labelled dose may achieve the same systemic exposure as a higher dose of isolate.
Epilepsy — The Largest Human Comparison (Pamplona et al., 2018)
The closest thing to a direct clinical comparison comes from a 2018 meta-analysis published in Frontiers in Neurology. Pamplona and colleagues analysed observational data from 670 patients with treatment-resistant epilepsy who received either CBD-rich cannabis extracts or purified CBD products.
The numbers were striking. Patients receiving CBD-rich extracts reported improvement at a rate of 71%, compared to 46% for purified CBD. The extracts required approximately 4× lower average daily doses — 6.0 mg/kg/day versus 25.3 mg/kg/day. Reports of adverse effects were also significantly lower with the extracts (Pamplona et al., 2018).
However, this finding comes with an important caveat. The purified CBD data came from controlled clinical trials (including the Epidiolex registration studies), while the extract data came from open-label observational studies — a significant methodological asymmetry. A 2023 scoping review in Cannabis and Cannabinoid Research specifically criticised this comparison, noting that observational designs are far more susceptible to placebo effects and reporting bias (Simei et al., 2023). The difference is suggestive, not conclusive. A head-to-head randomised controlled trial comparing full spectrum CBD oil to CBD isolate oil has not yet been conducted.
| Study | Year | What was compared | Full spectrum finding | Isolate finding |
|---|---|---|---|---|
| Gallily et al. | 2015 | Dose-response curve (inflammation) | Linear — effectiveness increased with dose | Bell-shaped — effective only at narrow dose range |
| Berthold et al. | 2023 | Oral bioavailability | 12–21% higher absorption | Baseline absorption level |
| Pamplona et al. | 2018 | Epilepsy outcomes (670 patients) | 71% improvement; 6.0 mg/kg/day dose; fewer side effects | 46% improvement; 25.3 mg/kg/day dose; more side effects |
| Raz et al. | 2023 | Terpene activity at CB1 receptor | Terpenes present in full spectrum activate CB1 at 10–50% of THC; 7 synergize with THC | No terpenes present |
The bottom line: Three independent lines of evidence — dose-response behaviour, bioavailability, and clinical outcomes — point in the same direction: full spectrum CBD may offer practical advantages over isolate. The evidence is strongest at the pharmacokinetic and preclinical level; a definitive head-to-head human trial has not yet been conducted.
The Science Behind the Difference — Why Does the Whole Extract Behave Differently?
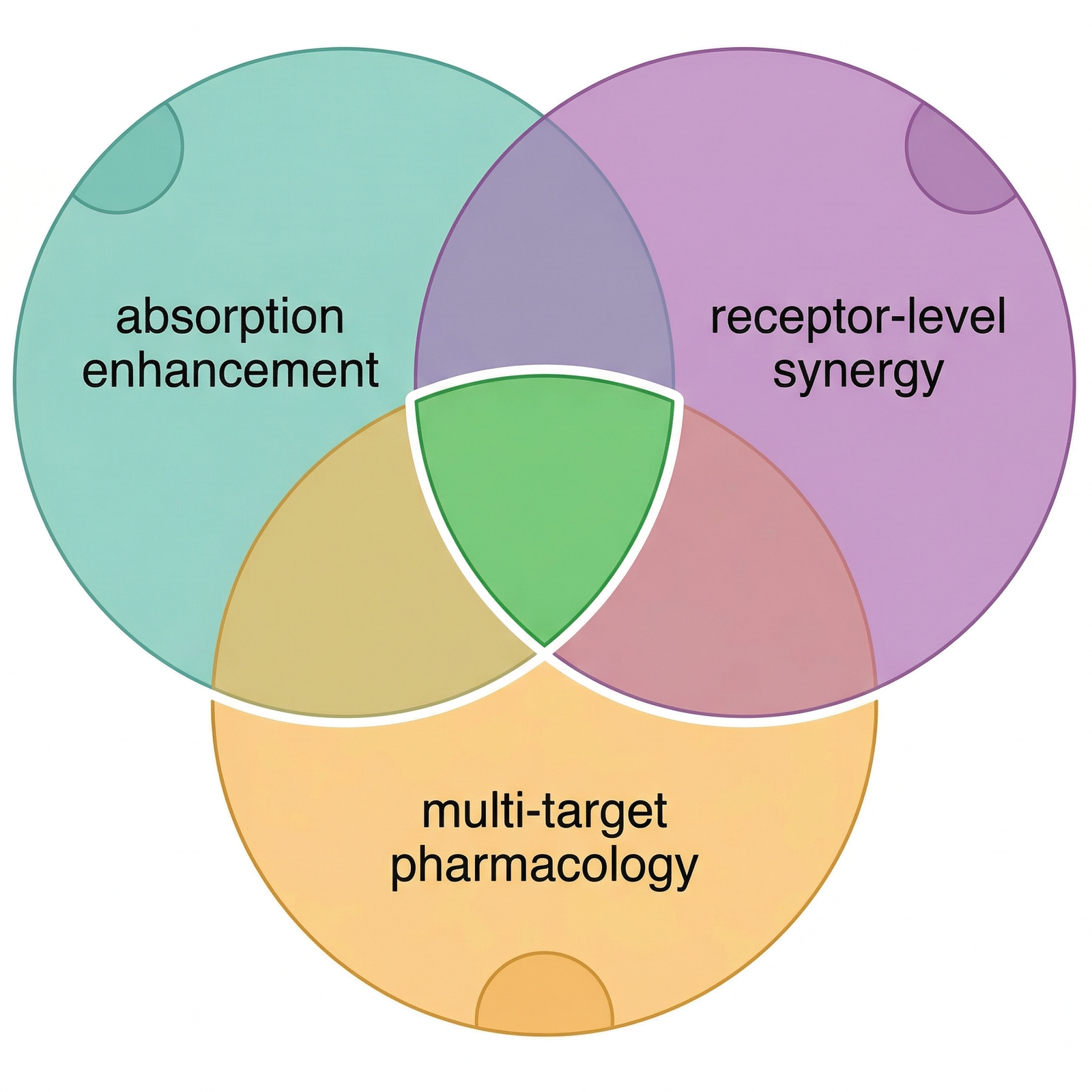
The data show a difference. But what is causing it? The answer is not a single mechanism — it is at least three, operating simultaneously.
First: pharmacokinetic synergy. The Berthold 2023 study identified a specific mechanism — trace THC increases CBD’s intestinal permeability while reducing its efflux through the gut wall. In practical terms, the presence of a small amount of THC helps CBD get through the intestinal barrier and into the bloodstream more efficiently. This is not a therapeutic effect of THC — it is a pharmacokinetic one, operating at the level of absorption.
Second: receptor-level synergy. The terpenes present in full spectrum extracts are not pharmacologically inert. The 2023 Raz et al. study demonstrated that cannabis terpenes activate CB1 cannabinoid receptors on their own, and that several — including limonene, linalool, and geraniol — synergize with THC at concentrations naturally found in the plant. β-Caryophyllene, typically the most abundant terpenoid in decarboxylated extracts, is a confirmed selective agonist at the CB2 receptor. In a full spectrum extract, these compounds contribute receptor activation that is entirely absent from an isolate.
Third: multi-target pharmacology. CBD itself interacts with over 65 molecular targets — far more than most pharmaceutical compounds. In a full spectrum extract, this already broad pharmacology is expanded further: minor cannabinoids like CBG act on different receptor subsets, terpenes contribute their own anti-inflammatory and anxiolytic properties through non-cannabinoid pathways, and the combined effect is a multi-target approach that may explain the more predictable dose-response observed by Gallily.
But does every combination produce synergy? This is where the nuance matters.
A 2024 systematic review in Pharmaceuticals tested several of the synergies originally proposed by Russo in his landmark 2011 review. Not all held up. The review found no evidence for neuroprotective effects of α-pinene and β-pinene against β-amyloid toxicity, and myrcene combined with CBD did not produce significant additional anti-inflammatory benefit (André et al., 2024). A 2023 scoping review in Biomedicines went further, arguing that the term “entourage effect” conflates several distinct pharmacological phenomena — synergy, bio-enhancement, polypharmacology — and that cannabis compounds can antagonise each other as well as cooperate (Christensen et al., 2023).
This does not invalidate the full spectrum rationale. It refines it. The evidence suggests that synergy is real but selective — certain compound combinations produce it, others do not. “Full spectrum” is not a guarantee of synergy; it is a starting condition that makes synergy possible. The quality of the specific extract — its terpene profile, its cannabinoid ratios, its processing — determines whether that potential is realised.
Why it matters: The full spectrum advantage is not one mechanism but at least three — improved absorption, receptor-level terpene activity, and multi-target pharmacology — working together. Not every compound combination produces synergy, but the evidence for specific synergies at the pharmacokinetic and receptor level is measurable and growing.
How to Evaluate a Full Spectrum CBD Product
Understanding the science is one thing. Evaluating a product on a shelf — or on a screen — is another. The research makes a compelling case for why full spectrum formulations may offer advantages. But “full spectrum” is a label, not a regulated standard, and the gap between what the term promises and what a specific product delivers can be significant.
The single most important tool for evaluating any CBD product is the Certificate of Analysis (COA) — a third-party laboratory report that details exactly what the product contains. A meaningful COA should show two things: the cannabinoid profile (how much CBD, how much THC, and whether minor cannabinoids like CBG, CBC, and CBN are present) and the terpene profile (which terpenoids are detected and at what concentrations).
Why does the terpene profile matter specifically? Because the pharmacological evidence discussed above — the Raz 2023 receptor data, the β-caryophyllene CB2 findings — depends on these compounds actually being present in the product you use. Extraction and decarboxylation involve heat, and heat drives off volatile monoterpenes like limonene and linalool. A product labelled “full spectrum” that shows no detectable terpenes on its COA has lost a significant part of what makes a whole plant extract pharmacologically interesting.
When reviewing a COA for a full spectrum CBD oil, look for the following: CBD content matching the label claim (within 10% tolerance), THC below the legal limit for your country (0.2% in most EU member states), at least two or three minor cannabinoids detected (CBG and CBC are the most common), and a terpene panel showing identified compounds — β-caryophyllene is typically the most resilient through processing and should be present in any genuinely full spectrum product.

As with any wellness decision, consulting a healthcare provider is recommended — particularly for those on existing medication, as CBD can interact with certain pharmaceuticals through cytochrome P450 enzyme inhibition.
One more consideration: trace THC. Full spectrum products in the EU contain less than 0.2% THC — far below any psychoactive threshold. However, with regular use at higher doses, even trace amounts can accumulate enough to produce a positive result on sensitive workplace drug tests. If drug testing is a concern, broad spectrum (THC-removed) or isolate products eliminate this risk, though they also remove the pharmacokinetic advantage that trace THC provides.
The choice between full spectrum, broad spectrum, and isolate is not about one being universally “better.” It is about which trade-offs align with your circumstances — your reasons for using CBD, your regulatory environment, whether drug testing applies, and how you respond individually. The evidence gives full spectrum a measurable edge in bioavailability and dose-response behaviour. Whether that edge matters for your specific situation is a decision only you can make. To explore full spectrum CBD options, look for products with transparent, third-party verified COAs.
What this means in practice: A COA showing both cannabinoid and terpene profiles is the best tool for verifying that a “full spectrum” product genuinely delivers the multi-compound profile the research describes — without it, the label is a claim, not a fact.
Regulatory note: EFSA’s February 2026 provisional safe intake level of approximately 2 mg/day for adults applies specifically to CBD preparations of ≥98% purity — effectively, CBD isolate. Full spectrum products, which contain multiple cannabinoids and terpenes alongside CBD, do not fall under this specific assessment and face distinct regulatory evaluation. No CBD novel food product — whether full spectrum or isolate — has yet received EU authorisation. Regulations vary significantly between member states. For a complete overview of CBD’s legal and regulatory status across Europe, see our dedicated guide: CBD Regulation in Europe: Current Status and What You Need to Know.
Frequently Asked Questions
Important: This article is for informational purposes only and does not constitute medical advice. CBD products are not medicines and are not intended to diagnose, treat, cure, or prevent any disease. Consult a healthcare professional before making decisions about your health. Regulations regarding CBD products vary across EU member states — read our complete guide to CBD regulation in Europe and verify the legal status in your country before purchasing.
Sources and Further Reading
- Gallily R, Yekhtin Z, Hanuš LO. Overcoming the bell-shaped dose-response of cannabidiol by using Cannabis extract enriched in cannabidiol. Pharmacology & Pharmacy. 2015;6(2):75–85.
- Berthold EC, Kamble SH, Kanumuri SRR, et al. Comparative pharmacokinetics of commercially available cannabidiol isolate, broad-spectrum, and full-spectrum products. European Journal of Drug Metabolism and Pharmacokinetics. 2023;48(4):427–435.
- Pamplona FA, da Silva LR, Coan AC. Potential clinical benefits of CBD-rich Cannabis extracts over purified CBD in treatment-resistant epilepsy: observational data meta-analysis. Frontiers in Neurology. 2018;9:759.
- Raz N, Eyal AM, Zeitouni DB, et al. Selected cannabis terpenes synergize with THC to produce increased CB1 receptor activation. Biochemical Pharmacology. 2023;212:115548.
- Simei JLQ, Souza JDR, Lisboa JR, et al. Does the “entourage effect” in cannabinoids exist? A narrative scoping review. Cannabis and Cannabinoid Research. 2023;9(5):1202–1216.
- Christensen C, Rose M, Cornett C, Allesø M. Decoding the postulated entourage effect of medicinal cannabis: what it is and what it isn’t. Biomedicines. 2023;11(8):2323.
- André R, Gomes AP, Pereira-Leite C, et al. The entourage effect in cannabis medicinal products: a comprehensive review. Pharmaceuticals. 2024;17(11):1543.
- Russo EB. Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. British Journal of Pharmacology. 2011;163(7):1344–1364.
- Gertsch J, Leonti M, Raduner S, et al. Beta-caryophyllene is a dietary cannabinoid. Proceedings of the National Academy of Sciences. 2008;105(26):9099–9104.
- EFSA. Provisional safe level for cannabidiol as a novel food. February 2026.